Universities in at least 19 states have reported outbreaks, despite health protocols on campus. Many outbreaks are tied to large group gatherings like parties, leading some schools to suspend students and organizations for breaking social distancing rules on and off campus.
The University of Notre Dame and the University of Alabama both have seen increases in Covid-19 cases on their campuses. Notre Dame has moved to online instruction, according to its website. Meanwhile, local and university police at the University of Alabama will partner to monitor bars, restaurants and off-campus housing to ensure the city’s Covid-19 ordinances and university guidelines are followed, university President Stuart R. Bell said.
“Violations to our health and safety protocols, both on and off campus, are subject to harsh disciplinary action, up to and including suspension from UA,” Bell wrote in a letter to the campus community on Sunday.
The University of Kentucky began a second phase of testing Sunday after a roughly 3% positivity rate for Covid-19 among fraternities and sororities in initial testing, which is triple the roughly 1% positivity rate for the general student population. And Central Michigan University has threatened to fine or suspend students who host large gatherings.
Penn State suspended its second fraternity this week for social distancing violations, according to a Sunday statement from the university.
Pi Kappa Alpha has been suspended for “hosting a large social gathering” on Saturday, that included about 70 students, the university said. The Phi Kappa Psi fraternity was previously suspended from the campus following an August 18 gathering that violated school policy, the university said.
Despite the rise in cases, University of Kentucky President Eli Capilouto cautioned against blaming students for the higher positivity rate on the school’s campus.
“Let me be clear: This is not an act to blame the students who reside in these facilities or who belong to these organizations,” he wrote. “We believe a number of factors associated with communal living spaces likely contributed to the high positivity rates in these residences.”
Across the US, there are 5.7 million cases of coronavirus and more than 176,000 people have died, according to the Johns Hopkins University database.
Concerns and questions about the new school year
On the K-12 front, school districts are still trying to figure out how to navigate the academic year during the pandemic.
Many schools across the country have implemented increased measures to protect students and staff against the virus, even though researchers are still learning how the virus spreads among young children.
The US Centers for Disease Control and Prevention updated its school guidance Friday emphasizing the importance of keeping schools open if possible. One big change in the guidelines was how schools can handle a positive case.
The CDC encouraged schools to work closely with local and public health leaders if there is an infected person on campus. But rather than shut everything down immediately for a long period of time, the guidelines said one option is an initial short-term class suspension and cancellation of events and after-school activities, so that public health leaders can get the time they need to determine how widespread the infections are.
If schools are using a pod system, keeping certain students together, administrators may only need to close certain parts of the building where an infected person had been.
A school district in Michigan canceled all classes and after-school activities for Monday — its first day of school — after “receiving a threat” on Sunday, according to an alert on the district’s website.
The Leslie Public Schools District did not offer details on the nature of the threat, but said it will work with law enforcement to assess the threat.
In Florida, a 6-year-old girl became the youngest person in the state to die from coronavirus complications. Health officials say they still don’t know if the child contracted the virus from a known case or if it was travel related.
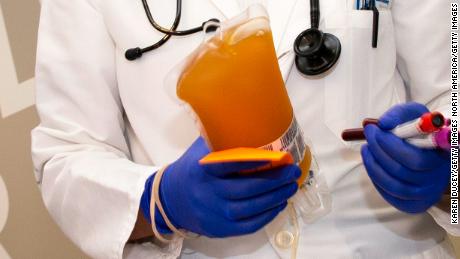
Convalescent plasma for Covid-19 treatment
The US Food and Drug Administration issued an emergency use authorization Sunday for convalescent plasma for the treatment of Covid-19 in hospitalized patients.
Convalescent plasma is created from the blood of people who have recovered from Covid-19, and it has shown some success in two other deadly coronaviruses: MERS and SARS. It has also been used to treat flu and Ebola.
The agency said it concluded that it may be effective in treating Covid-19 and that “the known and potential benefits of the product outweigh the known and potential risks of the product.”
Some experts, however, say there is not enough solid data to support the use of the plasma.
“The problem is, we don’t really have enough data to really understand how effective convalescent plasma is,” Dr. Jonathan Reiner, a professor of medicine at George Washington University and a CNN medical analyst said Sunday.
Art Caplan, founding head of the Division of Medical Ethics at NYU School of Medicine, told CNN he’s worried about whether there’s a large enough supply of convalescent plasma, which relies on donations from Covid-19 survivors.
“We’re going to get a gold rush towards plasma, with patients demanding it and doctors demanding it for their patients,” Caplan said.
Trump administration officials cited a Mayo Clinic-led study that showed a 35% improvement in survival among people given the highest doses of the treatment early on in their illness compared to those who were treated later.
FDA commissioner Dr. Stephen Hahn says studies have shown convalescent plasma is safe and the treatment has been given to patients with infectious diseases for more than a hundred years.
“There was a really good rationale for why this might work,” Hahn said at a White House briefing called in part Sunday to announce the emergency use authorization. “In the independent judgment of experts and expert scientists at FDA who have reviewed the totality of data … more than a dozen published studies … those scientists have concluded that Covid-19 convalescent plasma is safe and shows promising efficacy, thereby meeting the criteria for an emergency use authorization.”