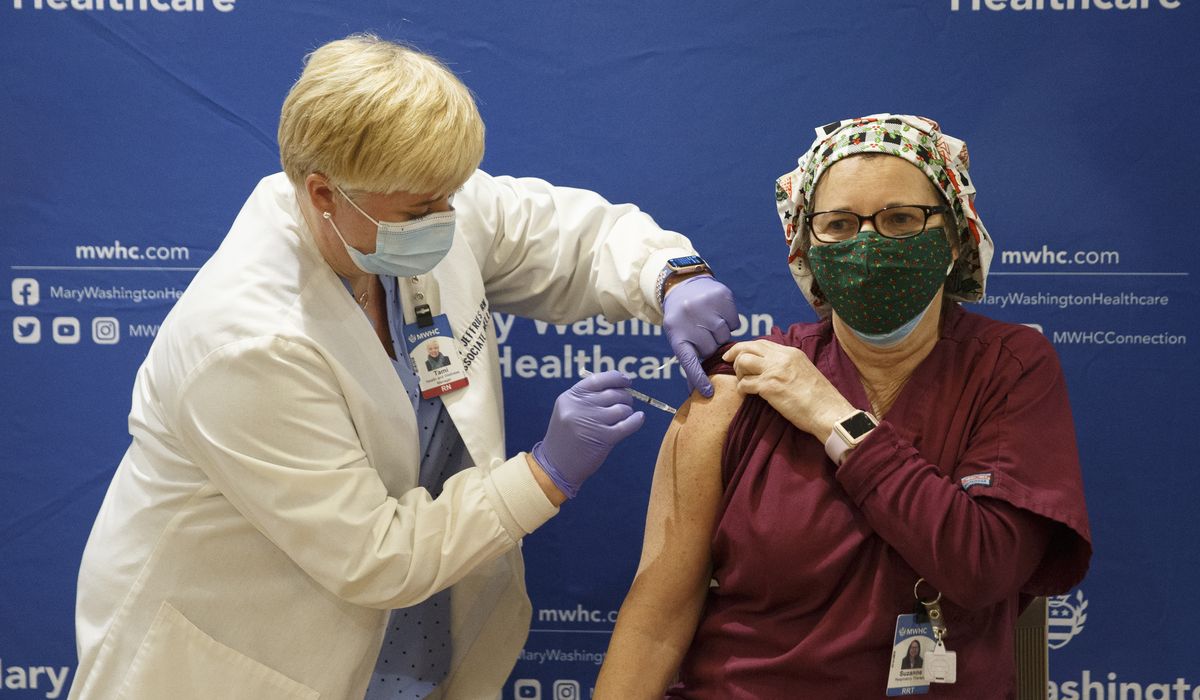
Health workers rolling up their sleeves across America are building confidence in a groundbreaking type of vaccine, one that uses “messenger RNA” to teach the body how to mount an attack in case the coronavirus comes along.
Doctors and nurses beaming for the cameras are being injected with snippets of genetic code that is protected by bubbles of fat known as a lipid nanoparticle.
Unlike most vaccines, which inject a weakened or inactive germ into the body, the shots from Pfizer and German company BioNTech spur the body to create replicas of the spike proteins that give the coronavirus its name.
“The body sees the spike, and says, ‘Uh oh, looks like a virus to me,’ and starts making a response to it,” Dr. Anthony Fauci, who leads infectious-disease research at the National Institutes of Health, told The Washington Times.
The technique has proven quite effective in trials, but surveys show at least a quarter of Americans are resistant to getting a COVID-19 vaccine, especially a new kind approved in record time.
Scientists say the steady drumbeat of doctors and nurses endorsing the technology with a shot to the deltoid will be a major boost.
“I think it’s extremely helpful. People still trust their doctors,” said Dr. Philip Dormitzer, Pfizer’s chief scientific officer for viral vaccines research and development. “Seeing health care providers, it is a real testimonial. These people, they’re putting their arms where their mouth is — which is a funny way of saying it.”
The Food and Drug Administration on Tuesday released data affirming that a second mRNA vaccine, from Moderna, is nearly 95% effective against COVID-19, putting it on par with the Pfizer shots.
Moderna’s vaccine is on track for approval by Friday, giving the U.S. a second stream of vaccines to protect frontline doctors and nurses and nursing-home residents in the initial phase of the rollout. Federal officials say they can get shots into 20 million people before the end of the year.
Vice President Mike Pence on Tuesday toured the Catalent manufacturing plant in Indiana that is filling and packaging doses of the Moderna vaccine.
Speaking at a roundtable, Indiana Gov. Eric Holcomb told Mr. Pence that footage of early vaccinations reminded him of the part of “O Holy Night,” that goes, “A thrill of hope, the weary world rejoices.”
“I think that’s what Hoosiers and Americans are feeling right now. We can feel the light,” he said.
The mRNA vaccines rolling off the assembly line are the frontrunners in a broader portfolio from President Trump’s vaccine initiative, Operation Warp Speed, which tapped six drugmakers to develop vaccines under three platforms.
To hedge their bets, two companies worked within each platform. Besides the mRNA pair, AstraZeneca and Johnson and Johnson are working on vaccines that use an adenovirus vector, while Novavax and the Sanofi-GlaxoSmithKline partnership are using recombinant proteins.
Moderna and its government partners at NIH designed a vaccine within days of receiving the genetic sequence of the coronavirus from China in January, reflecting the agility of the mRNA platform.
“You just pull the code out, you synthesize that bit of RNA and you’re good to go,” Dr. Fauci said.
The technology is decades in the making, but the COVID-19 crisis is its first real test. The stakes couldn’t be higher, and data from phase 3 trials of the Pfizer and Modern vaccines wowed the scientific community.
“There were people who were skeptical, saying, ‘Why are you making it one of the main tools in this important, historic pandemic?” Dr. Fauci said. “Sure enough, we were right.”
Pfizer began working with German company BioNTech, a leader in developing mRNA therapies, in 2018 on a flu vaccine. But they pivoted to COVID-19 to address the swirling crisis that has killed more than 302,000 people in the U.S.
Dr. Dormitzer said when he heard about mRNA technology earlier in his career, “I was skeptical about this.”
“As we started to gain more and more experimental data, it kept on looking good,” he said.
The vaccines’ potential could help federal and state officials convince more people to roll up their sleeves and contribute to the herd immunity needed to break the pandemic.
The Kaiser Family Foundation on Tuesday said 12% of Americans say they probably won’t roll up their sleeves and 15% say they definitely won’t, with Republicans, rural residents and Black adults the most leery.
Foundation President and CEO Drew Altman said those attitudes might change as the immunization campaign plays out, especially if trusted doctors and federal officials give the vaccines their stamp of approval.
“Many who are hesitant are in wait-and-see mode,” he said, “and their concerns include worries about side effects and whether the vaccine can cause COVID-19, which may dissipate as people get more information and see the vaccine introduced successfully among people they know.”